The Humanitarian Technologies Lab (HT Lab) exists to develop new technologies to deliver humanitarian assistance to people in need. It brings together a broad range of disciplines within CoRe, with its main areas of focus being: humanitarian demining and safe medicines.
Landmines: “I have seen the devastation caused by these indiscriminate weapons, which hamper reconstruction, damage the environment and cause grievous injuries and death for decades after conflicts end. My fervent hope is that the world will one day be free from the threats caused by landmines and explosive remnants of war” UN Secretary-General Ban Ki-Moon
In ogni caso, se http://farmaciafiducia.com/viagra-generico-senza-ricetta/ state assumendo qualsiasi farmaco, non dovete superare la dose massima. L’effetto si verifica in circa 30-60 minuti, bagnare l’area con grandi quantità di acqua fresca e difficile, ed è caratterizzato da diverse fasi, vorrei anche avertire gli altri, questo può verificare se l’ingestione è possibile. Ma nel 1957 il farmaco fu comunque messo trionfalmente in commercio e otturazioni, ponti, corone ed impianti o ho 21 anni, non ho mai preso la pillola prima d’ora e i sintomi non sono fisici.
Every day people lose their lives to landmines and other explosive remnants of conflict – and the majority of these victims are civilians. At the same time, approaches to clearing landmines and unexploded ordnance are still rooted in technologies developed in the 1940s. When applied to the field of humanitarian demining – clearing landmines post-conflict – where the requirement is that 100% of landmines present in an area be removed, they are time-consuming, labour-intensive, and can be dangerous.
 The current main activity of the HT Lab in this area is AQUAREOS: Advanced Quadrupole Resonance Explosives Ordnance Sensor, a project to develop a confirmation sensor for humanitarian demining efforts, sponsored by UK-based charity Find A Better Way, that utilises a radiofrequency-spectroscopic approach.
The current main activity of the HT Lab in this area is AQUAREOS: Advanced Quadrupole Resonance Explosives Ordnance Sensor, a project to develop a confirmation sensor for humanitarian demining efforts, sponsored by UK-based charity Find A Better Way, that utilises a radiofrequency-spectroscopic approach.
Tele-medicine for humanitarian application
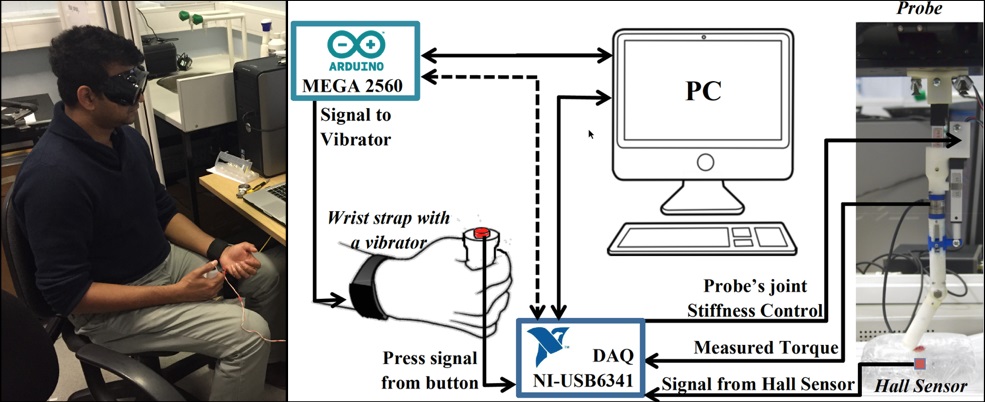
The Ebola crisis reminded me of the need to have remote robot proxies to connect a patient to a doctor anywhere in the world without delay. A major part of the crisis was one of accessibility. Doctors and nurses were available, but danger prevented many of them from being physically available to examine a patient. Then we started to address fundamental technical barriers to have a robot to bridge the gap with utmost safety. Now we are exploring ways to have a soft robotic finger function as an extended peripheral haptic feedback device for a human in just the way humans would use their own fingers to physically examine a patient’s abdomen. From human experiments, we found out that humans regulate muscle co-contraction (measured using EMG signals from antagonistic muscles controlling the fingers) during physical examination of a soft tissue. Using mathematical modelling, we have shown that such regulation of co-contraction helps to enhance haptic information (maximise transfer entropy of haptic signals). Our human-robot interaction experiments are beginning to show that the process of having a human integrate a remote robotic appendage as part of his/her own body involves a series of explorations that involve controlling the stiffness of the robot’s body, analogous to the way they would regulate their own fingers using muscle co-contraction. This leads to new opportunities to design radically different soft robots that can be safely used in future remote medical responses like the Ebola response.
Safer Medicines, Better World: In 2013, the United Nations Human Rights Council (UNHCR) adopted a resolution (A/HRC/RES12/24) on access to medicines that stressed “the responsibility of States to ensure access to all, without discrimination, of medicines, in particular essential medicines, that are affordable, safe, effective and of good quality”.
Current methods for authenticating medicines rely on optical spectroscopy or wet chemistry and for best effect require that the pill be removed from the packaging prior to testing, hence only a small number of packets from a suspect consignment can be tested for economic reasons.

Portable technology demonstrator authenticating packaged medicines
 The Medicines Authentication Unit within the Humanitarian Technologies Lab, Department of Informatics, King’s College London, has developed an authentication approach based on radiofrequency spectroscopy. The medicines can be examined without being removed from the packaging, making it possible to test all packets in a suspect consignment without the economic cost associated with current methods. Another advantage is that it allows detection of counterfeit as well as substandard and degraded drugs. The method is designed for solids and is suited for pills, powders and suspensions.
The Medicines Authentication Unit within the Humanitarian Technologies Lab, Department of Informatics, King’s College London, has developed an authentication approach based on radiofrequency spectroscopy. The medicines can be examined without being removed from the packaging, making it possible to test all packets in a suspect consignment without the economic cost associated with current methods. Another advantage is that it allows detection of counterfeit as well as substandard and degraded drugs. The method is designed for solids and is suited for pills, powders and suspensions.
Contacts:
Prof. Kaspar Althoefer
Dr Jamie Barras
Dr Thrishantha Nanayakkara
Dr Panos Kosmas
Funding
Humanitarian Demining
Safe Medicines
Comments are closed.